Market Ready Liposomal Products
Structured delivery. Controlled manufacturing. Accelerated time to market.
High-quality, stability-tested, proven liposomal formulations available for rapid commercial launch
Ready to Market Products
Manufactured by liposomal specialists

A streamlined route to market
GMPriority Pharma provides white label liposomal products for brands seeking high-quality, stability-tested formats, manufactured within controlled GMP environments.
By aligning formulation design, processing parameters and manufacturing conditions from the outset, products are built to perform consistently beyond the laboratory, supporting efficient launch and dependable supply.
This approach reduces development timelines while maintaining control over product quality, stability and performance.
What's Included
White label programmes are delivered within a structured manufacturing framework, providing:
- Pre-developed liposomal formulations
- Manufacturing within GMP-aligned environments
- Defined processing and batch control
- Scalable production pathways
- Ongoing supply capability
- Standard technical documentation
What makes GMPriority Pharma different
Unlike generic white label products, GMPriority Pharma formulations are developed using a delivery-first framework that aligns lipid structure, particle behaviour and processing conditions.
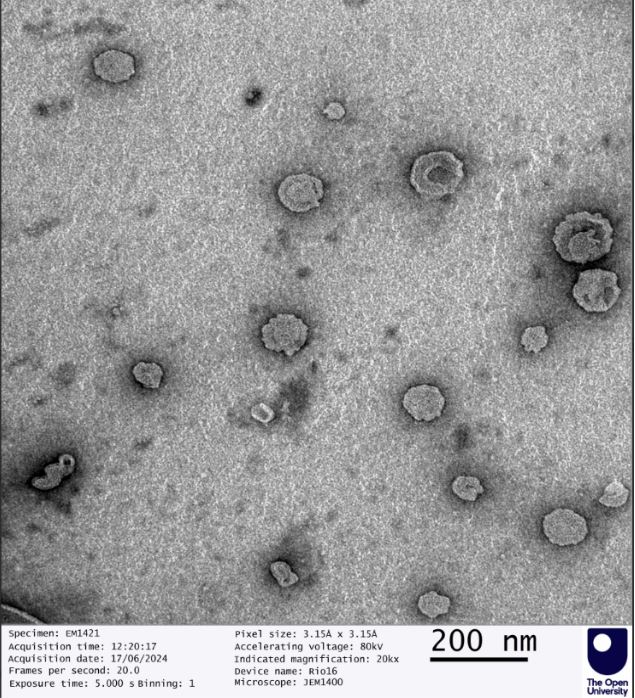
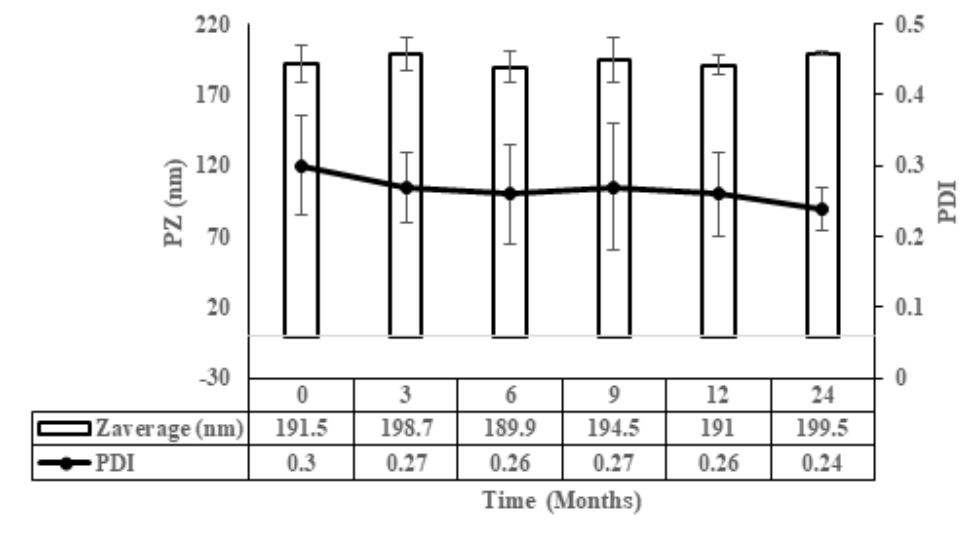
Liposomal systems are sensitive to formulation and processing variables. White label products from GMPriority Pharma are developed with defined control over these parameters to provide consistent particle size distribution, stable dispersion behaviour and reproducible batch output.
This enables reliable product performance across manufacturing cycles and future supply batches.
Built for Repeatable Performance
Example Actives
- Vitamins
- Minerals
- Plant Compounds
- Antioxidants
- Lipids
- Combination Formulations
Available in liquid, capsule and powder format.
Availability and minimum order volumes vary by product category and packaging configuration.


Manufacturing Standards
All white label products are produced within GMP-certified, FSSC ISO 22000-accredited facilities, with defined process control and quality frameworks.
Each production batch follows strict processing parameters to maintain consistency in particle distribution and structural stability.
Why White Label?

White label is suitable for:
- Established supplement brands expanding into liposomal formats
- Distributors entering the liposomal category
- Emerging brands requiring technical manufacturing support
- Companies prioritising speed to market with controlled quality
From Manufacturing to Market
This route allows brands to focus on positioning, marketing and distribution, while GMPriority Pharma manages liposomal manufacturing within defined quality parameters.
Structured production process
Defined quality systems
Reliable batch manufacturing
Technical transparency
Commercially Structured for Growth
White label programmes operate within defined commercial frameworks, supporting:
- Production scheduling visibility
- Structured supply agreements
- Scalable order volumes
- Ongoing manufacturing capability
This ensures clarity from launch through to sustained market supply.
Launch your product with confidence
GMPriority Pharma’s white label programmes provide a structured, controlled route to market, combining delivery science, manufacturing discipline and commercial readiness.